
Is Hexane Polar or Nonpolar Nehemiahrilrexton
Is IF5 polar or non polar? Updated: 8/11/2023 Wiki User ∙ 9y ago Study now See answers (2) Best Answer Copy IF5 is considered a type of polar molecule. It is a polar molecule because it.

Is IF5 Polar or Nonpolar? Techiescientist
There are four electron groups around the central atom. As shown in Figure 9.2.2 9.2. 2, repulsions are minimized by placing the groups in the corners of a tetrahedron with bond angles of 109.5°. 3. All electron groups are bonding pairs, so the structure is designated as AX 4.
PPT Part 09 IMFA and Solubility PowerPoint Presentation, free
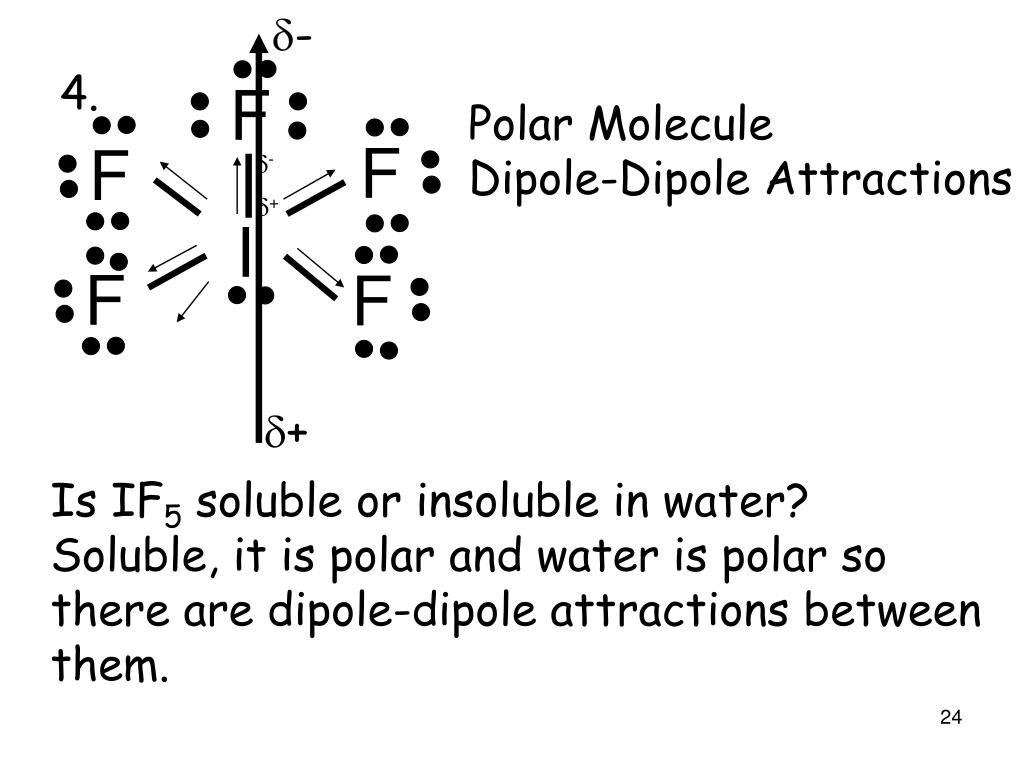
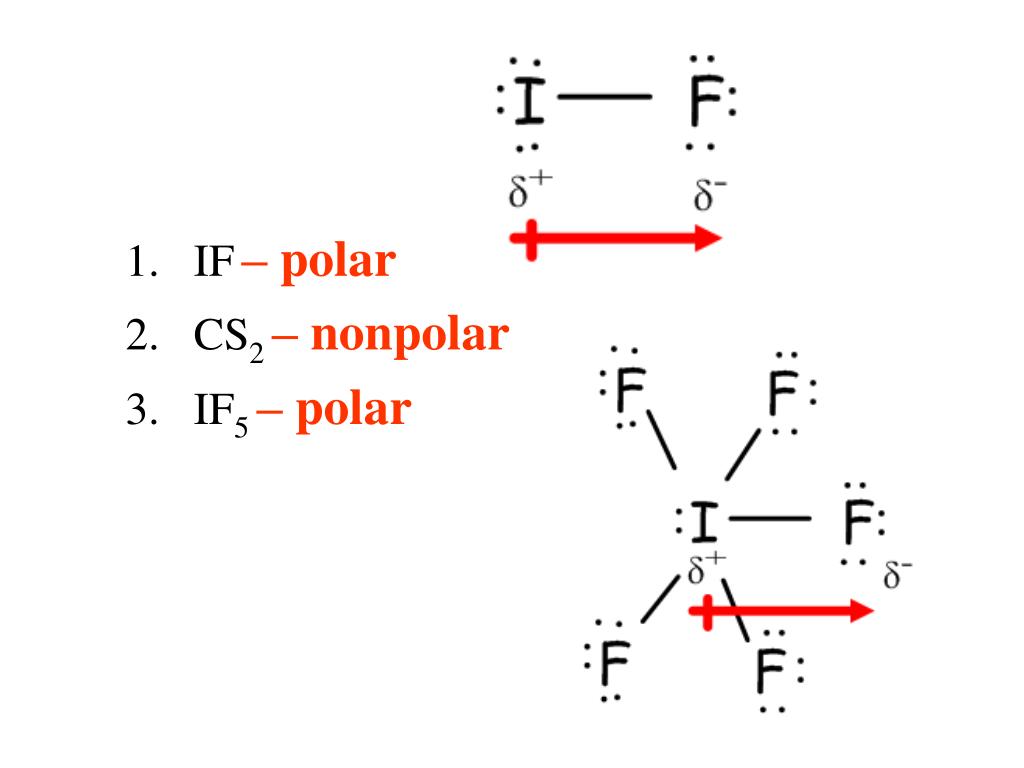
Chemistry Chemistry questions and answers Specify whether the molecule IFs is polar or nonpolar and explain why. The molecule is polar because all the I-F bonds are polar and the net dipole moment is nonzero. The molecule is polar only because all the I-F bonds are polar. The molecule is nonpolar because all the I-F bonds are nonpolar.
PPT Unit 79 Test Review PowerPoint Presentation, free download ID
Guide Is If5 Polar Or Nonpolar September 22, 2022 Webster West The Iodine pentafluoride chemical formula is IF5. Drawing IF5 Lewis Structure is very easy to by using the following method. Here in this post, we described step by step method to construct IF5 Lewis Structure.
What Is The Difference Between Lewis Structure And Vsepr
a. SiCl4 b.. | Channels for Pearson+ Next General Chemistry 13. Liquids, Solids & Intermolecular Forces Molecular Polarity 7:17 minutes Problem 52 Textbook Question Determine whether each molecule is polar or nonpolar. a. SiCl4 b. CF2Cl2 c. SeF6 d. IF5 Verified Solution 7m

Polar and Nonpolar Molecules YouTube
Molecules can be classified as polar or nonpolar. The molecule polar behaves in a different manner as compared to nonpolar. Overview: IF5 Lewis Structure. The central atom is Iodine, which is bordered on five terminals with fluorine atoms( in square pyramidal), and one lone pair on the central in the square pyramidal geometry.
Is BrCl5 Polar or Nonpolar? (Bromine Pentachloride) YouTube
The molecule is symmetric. The two oxygen atoms pull on the electrons by exactly the same amount. Propane is nonpolar, because it is symmetric, with H atoms bonded to every side around the central atoms and no unshared pairs of electrons. Exercise 4.12. 1. Label each of the following as polar or nonpolar.
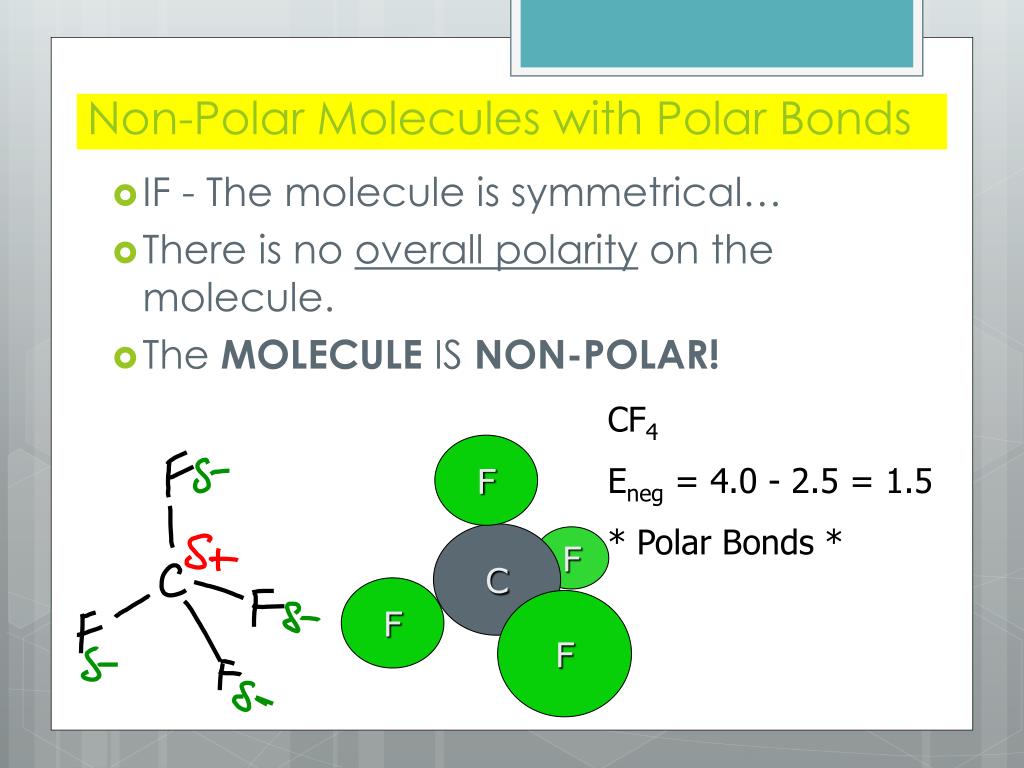
Cf Polar Or Nonpolar Slidesharetrick My XXX Hot Girl
So, Is IF5 Polar or Nonpolar? IF5 is polar in nature. The molecule has a bent shaped geometrical structure because of lone pair and bond pair repulsion as per VSEPR theory due to which there occurs an imbalance in charge distribution across the molecule.

If5 Lewis Structure
Henry Agnew (UC Davis) 5.10: Electronegativity and Bond Polarity is shared under a not declared license and was authored, remixed, and/or curated by LibreTexts. Covalent bonds can be nonpolar or polar, depending on the electronegativities of the atoms involved. Covalent bonds can be broken if energy is added to a molecule.
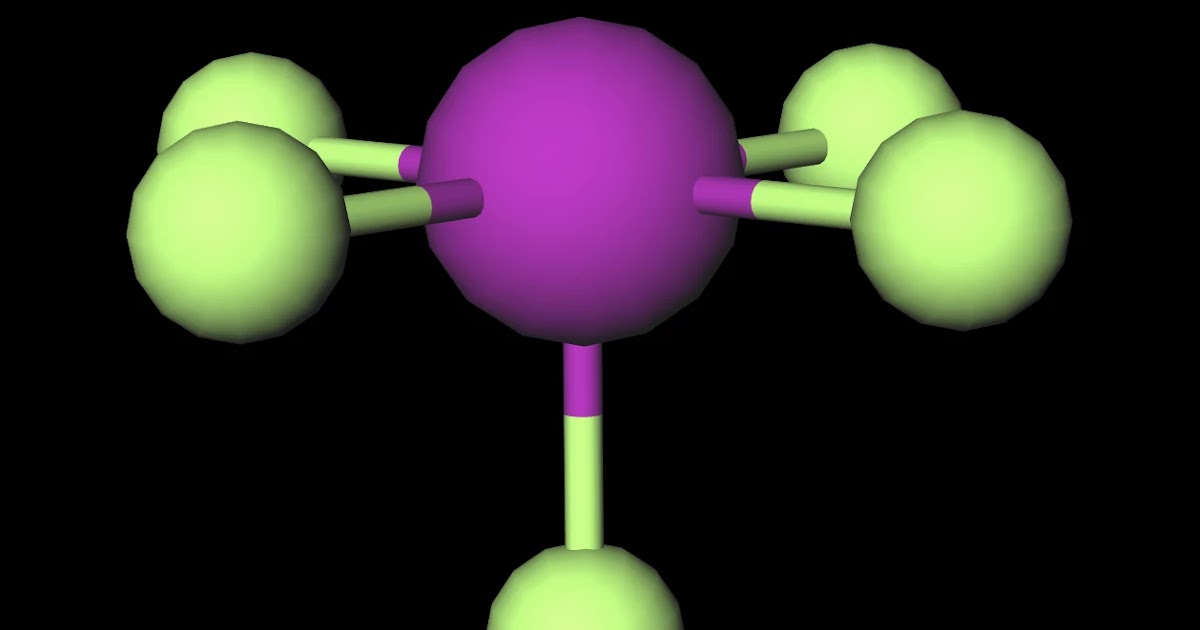
MakeTheBrainHappy Is IF5 Polar or Nonpolar?
Is IF5 Polar or Nonpolar? Answer: IF5 is a polar molecule due the presence of a lone pair of electrons which due to electron-electron repulsion results in a bent structure. This leads to an unequal distribution of charge within the molecule and therefore a permanent dipole.
[Solved] image attached 1. Complete the table below. Indicate whether
1. The first step is to count all the valence electrons of each molecule. In the case of IF5, The Iodine atom has 7 valence electrons. F also has 7 valence electrons. But since there are 5 atoms of F, we multiply 7×5= 35 valence electrons. Adding both we get 35+7= 42. Hence, a total number of valence electrons of IF5= 42.

Polar Vs Nonpolar Ciencias exatas, Ciência química, Físicoquímica
Iodine pentafluoride (IF5) is a polar molecule. The central iodine (I) atom in IF5 is surrounded by five fluorine (F) atoms forming a square pyramidal shape. The electronegativity of the fluorine (F) atom is greater than the iodine (I) atom.

Polar vs. Nonpolar Bonds — Overview & Examples Expii Ionic Bonding
The bond polarity between two atoms can be estimated if you know the electronegativity of both elements. Calculate the electronegativity difference (ΔEN) and average ( EN) of the two electronegativities, and use the table below to determine the bond type and polarity. Calculate the molecular polarity (polar, non-polar) of a chemical bond based.

Is HF Polar or Nonpolar? (Hydrofluoric Acid) YouTube
BrF3, or bromine trifluoride, is a powerful fluorinating agent for chemical reactions with sp3d hybridization in its center bromine atom. It's a T-shaped molecule with an 86.2° bond angle. The molecule is very polar, and it is mostly utilized to make uranium hexafluoride during uranium processing.
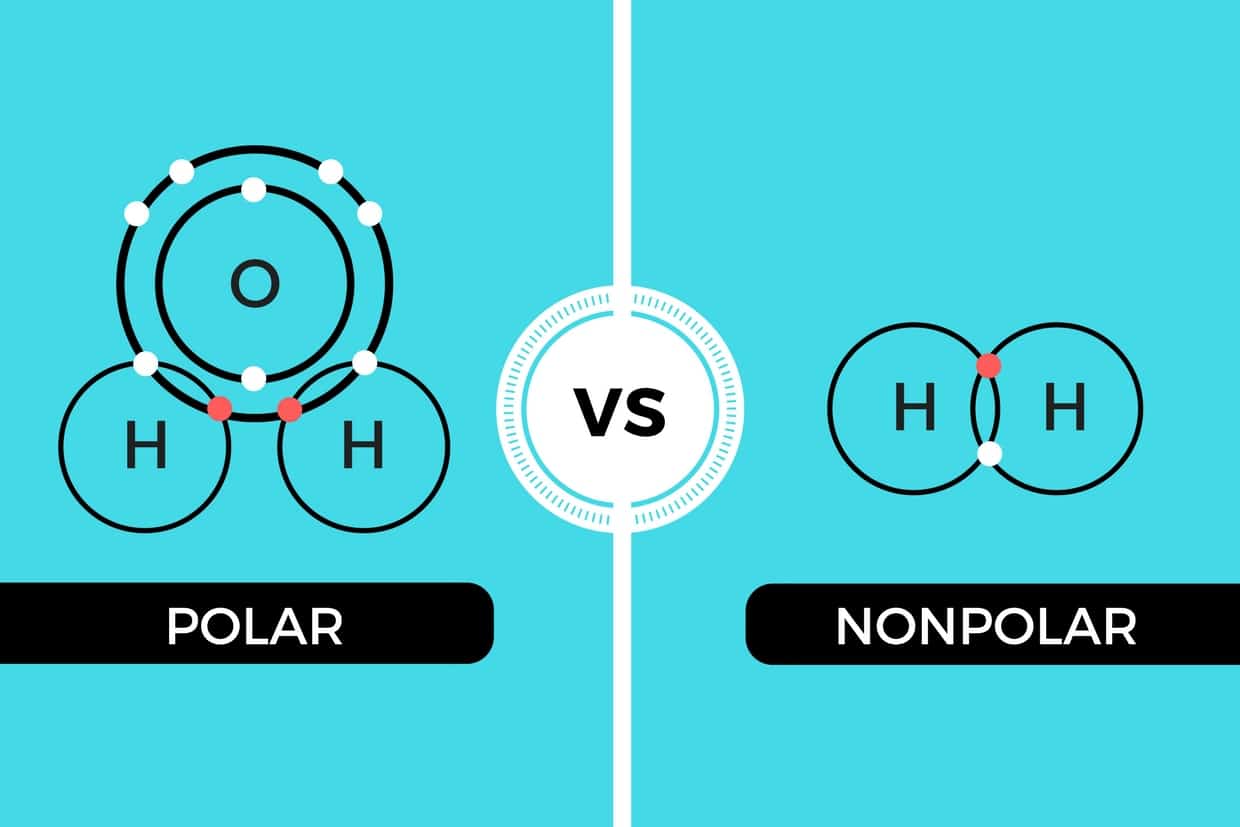
Difference between polar and nonpolar examples
IF5 has a molecular mass of 221.9 grams per mole and a density of 3.25 g/cm3. It is a melt temperature of -28.8degC, and the boiling point is 47.3degC. The IF5 molecule is polar because of its asymmetrical structure. It comprises an iodine atom centrally bonded to five fluorine molecules in a bipyramidal trigonometric arrangement.

Is IF5 Polar or Nonpolar? Techiescientist
100% (11 ratings) Part k The molecule. View the full answer Transcribed image text: Part A Draw an appropriate Lewis structure for IFS. Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone Q (5 ଡ BO Identify the geometry of IF5 using VSEPR theory.